How Does Global Warming Work?
By Chris Tilley
I was recently asked how global warming works, in particular what carbon dioxide has to do with it. The simple answer is that the carbon dioxide acts as a blanket keeping the heat in. The more carbon dioxide, the heavier the blanket. The more complex answer involves global energy budget and absorption bands.
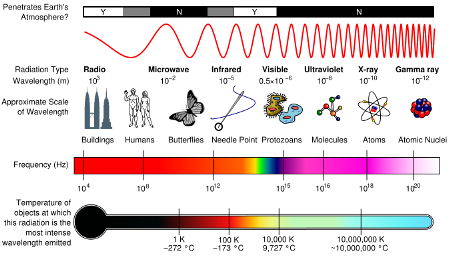
First up is where earth gets its energy. The vast majority (99.978%) of it comes from the sun. The tiny remainder comes from geothermal, tides and our fossil fuel consumption.(source) So what actually comes out of the sun? Electromagnetic radiation of all wavelength of which visible light is a tiny sliver.
Fortunately for us most of those wave lengths don’t get through our atmosphere.
Source (Wikipedia)
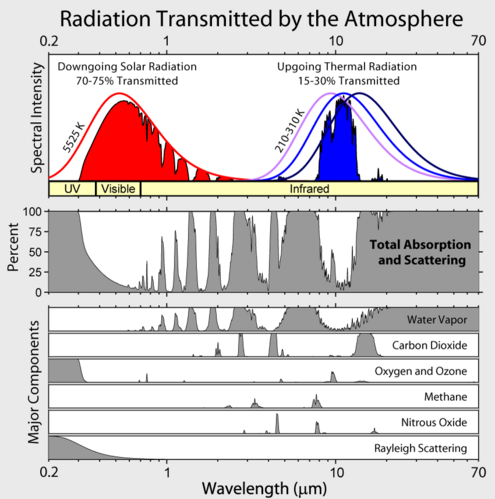
So what does get through?
Source (Wikipedia)
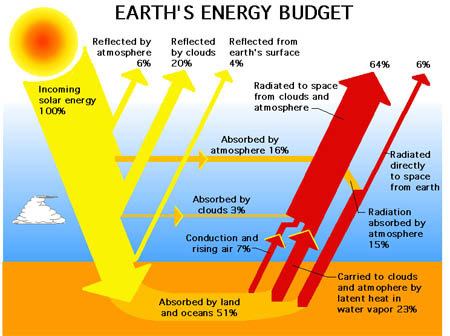
Coming down mostly visible light with some UV and infrared. Of that light 30% is reflected back into space by the atmosphere, clouds and the ground. As an interesting side note melting ice will decrease the amount of light reflected and more clouds would increase the amount reflected. The remaining light is absorbed and heats. For and example hold you hand out in the sun, you can feel your hand getting warm from the sun. Next hold your hand over a car that has been out in the sun for a while and you will feel the heat radiating off of it.
The heat radiating off the car is in the infrared range of the spectrum. That also transmits through our atmosphere but here 70% makes it out to space. The remaining 30% provides the energy for our weather.
Source (Wikipedia)
Going back up to the absorption image we can see that what keeps the infrared wavelength in is carbon dioxide and water vapor as well as some other gases. So why do certain wavelengths pass through and others don’t?
First, all electromagnetic radiation consists of photons. The photons have a certain amount of energy and that is directly related to the wavelength. So a photon with a certain energy level may be in the visible wavelengths, where one with more energy could be in the ultraviolet wavelengths. Atoms and molecules can absorb a photon in certain bands if the energy the photon has exactly matches what the atom needs. What the atom or molecule needs is based on its internal structure. The exact details of how this works can be found at wikipedia. The absorption bands of carbon dioxide are mostly in the infrared as can be seen in the above image.
So the result is that carbon dioxide acts as a blanket for the earth by letting visible light in and trapping some of the infrared going out. Increasing carbon dioxide will act as a heavier blanket keeping more infrared in. The opposite is also true without any carbon dioxide in the atmosphere there would be no liquid water on the earth with a temperature of -18 C. That compares to the actual value of 14 C.(source) So we need some but to much will raise temperatures.